Super User
Embryo Cryopreservation
Embryo Cryopreservation is the process by which the surplus embryos from an IVF or ICSI cycle may be stored or frozen, for use at a later date.
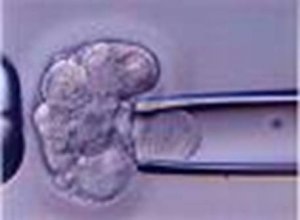
Embryos can be frozen at a number of their developmental different stages – up to and including blastocyst stage. The Embryologist selects the embryos suitable for freezing. The embryos are then introduced into special culture media, called cryoprotectant, to help protect them during the freezing and storage process. The embryos are then frozen using a computerised embryo freezer.
Once frozen, the embryos are contained within appropriately labelled sterile straws. These straws are then securely stored in a locked storage vessel immersed in liquid nitrogen ( - 196 C). Embryos can be safely stored like this for up to 5 years.
Embryo Thawing is the process by which frozen embryos are thawed with a view to replacing the most suitable into the woman’s uterus. The freezing and thawing of embryos allows patients use surplus embryos from previous cycles.
During thawing, the frozen embryos are warmed in a controlled manner. Once thawed the embryos are introduced back into embryo culture. The embryos are thawed the day before the embryo transfer in order that the embryo’s development can be monitored. Not all embryos survive the freeze/thaw process.
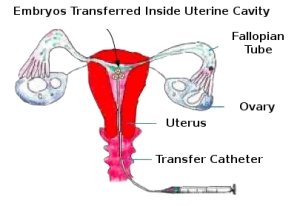
Our Fertility Specialist performs the embryo transfer procedure. The selected embryos are introduced, through a fine catheter, into the woman’s uterus. Typically 2-3 embryos are transferred, depending on the patient’s circumstances. The embryo transfer procedure takes no more than half an hour and few women experience any discomfort.
Again, the spare embryos are then assessed in terms of their suitability to be re-frozen for use, by the couple, in future cycles.
Worldwide, the success rates for frozen-thawed embryos are lower than those for fresh embryo transfers.
Vitrification
Having the ability to preserve embryos is important service to our patients. Fertility units, worldwide, have used the same "freezing" technique for the last 25 years - with little modification.
One of the major huddles of "freezing" is that ice crystal formation is VERY damaging to embryos - this hurdle had never really been overcome. This is one of the reasons that, even the most capable IVF Units, rarely achieve that same results with frozen embryos as they were getting with fresh embryos.
Vitrification works on a completely different principle for cryo-storing embryos. Vitrification addresses the problem of ice crytal formation and allows for a reliable and successful cryopreservation of your embryo(s). Vitrification is a "glass like" state in which the embryo is suspended among high concentrations of cryoprotectants in liquid nitrogen.
The embryo survival rates are fanatastic and this has led to our "frozen" embryo pregnancy rates being almost equal to those of our fresh embryo transfers.
PGS - Preimplantation Genetic Screening
is a procedure that involves biopsying the embryos and identifying which of the embryos is at risk of a specific genetic disease, such as cystic fibrosis.
Pre-implantation genetic screening (PGS) can also be used to screen for embryos with an abnormal number of chromosomes –with a view to increasing the likelihood of an ongoing pregnancy. The main indications for PGS are an advanced maternal age, a history of recurrent miscarriages or repeated unsuccessful implantation. It has also been proposed for patients with obstructive and non-obstructive azoospermia.
One or two cells are biopsied from the embryos and then flown to a world-leading laboratory in the United States. They perform the genetic analysis and get the results back to us within 24 hours –and this aids in selecting which embryos are likely to be free from the genetic disease
After the embryo replacement, surplus good quality unaffected embryos can be vitrified for use in future / sibling cycles.
This represents a significant coming together of state of the art technologies :
Micromanipulation techniques and technology which enable ICSI to create the embryos and to then perform the embryo biopsy
Genetic Screening – to help in selecting which embryos are likely to be free from the genetic disease.
Vitrification – to store surplus embryos are likely to be free from the genetic disease for future use and siblings.
Testicular Biopsy (TESE)
Testicular biopsy is used to collect sperm directly from the testis or the tubes conveying sperm from the testis. The testis might be creating sperm, which may not be present in the ejaculated semen. This may be for a number of reasons such as:
- An absence of the tubes that convey the sperms from the testis –this may due to having had a vasectomy or it can occur in patients with cystic fibrosis.
- A blockage of the tubing –this may be due to infection.
There are two common approaches to testicular biopsy:
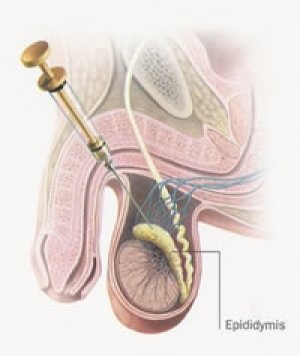
Percutaneous Epididymal Sperm Aspiration (PESA).
PESA is when a fine needle is inserted into the tubes that convey the sperm out of the testis. One area of this tubing, the epididymus, is a natural reservoir for sperm and is therefore a good place to aspirate them from.
Testicular Biopsy.
If a PESA is not possible or no sperm are identified then the procedure progresses onto testicular biopsy. This procedure involves the removal of very small pieces of tissue from the testis –the Embryologist then processes the biopsies with a view to finding motile sperm.
The testicular biopsy procedures are performed under local anaesthetic. The procedure takes about 15 minutes.
Sperm suitable for ICSI are sometimes not found. It is prudent, therefore, to have some form of sperm as “back-up” in order that the ICSI treatment cycles not have to be abandoned.
The sperm recovered by either of these techniques is only suitable for use with ICSI. Once the ICSI has been completed and sperm remaining, of good quality, may be frozen for use with future ICSI cycles.
The success rate for ICSI is typically 30 – 55% per treatment cycle. However, the success rate is dependant on many factors, such as the age of the woman.
Blastocyst Transfer
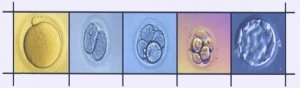
In natural conception embryos arrive in the uterus 5-6 days after fertilisation. With present day IVF, embryos are routinely transferred back 3 days after fertilisation. Recent breakthroughs in the laboratory and developments in media culture have made it possible to grow embryos in vitro for up to 5 days. This is referred to as blastocyst development.
By growing the embryos to blastocyst stage you have the potential to choose the best developing embryos and also to transfer fewer embryos, which results in increased pregnancy rates and lowers the multiple pregnancy rates.
However, not all patients are suitable candidates for this procedure.
Assissted Hatching
Why Perform Assisted Hatching?
There is another critically important component of the embryo that is often overlooked, the zona pellucida (ZP) or the egg shell. The zona pellucida is secreted by the egg and has several important functions, such as:
i) During fertilization, it serves to prevent the access of more than one sperm to the egg.
ii) it keeps the cells of the embryo together during early development, until the embryo reaches the blastocyst stage. At the blastocyst stage, the embryo has enough structural integrity that it no longer needs the protection of the ZP and the embryo “hatches”. A higher implantation rate is observed in patients undergoing IVF, who had the ZP of their embryos mechanically opened.
Who Needs Assisted Hatching ?
The following patient groups may benefit from assisted hatching : women older than 36 years of age, those with elevated FSH levels, couples with previous IVF failures, embryos with an abnormal appearing zona pellucida, and when using cryopreserved embryos.
How is Assisted Hatching Performed?
There are a number of ways to introduce a hole or a weakness into the zona.
Mechanical: Some embryologists will perform assisted hatching by mechanically piercing the ZP with a specifically designed pipette.
Chemical Erosion: involves exposure of the embryo to an acidified media called acidified tyrodes solution. This acidic solution is microscopically applied to the ZP to induce thinning.
Laser: This specifically designed laser system includes the laser, which serves as the energy source to create an opening in the ZP, and a computer which allows the operator to precisely control the laser energy output, laser pulse duration, and gap size. This system has many benefits when compared to the more standard mechanical and acidified tyrode’s techniques.
The computer assisted laser method is more precise, resulting in a more consistent ZP opening. Laser assisted hatching takes less time and does not expose the embryos to potentially adverse chemicals, so embryos spend less time out of the optimal culture conditions of the incubator.
The cumulative effect of these factors is to minimize the stress placed upon the embryo during the performance of assisted hatching. This in turn translates into improved implantation and pregnancy rates.
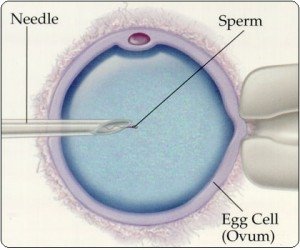
Intra-Cytoplasmic Sperm Injection (ICSI)
Intra-Cytoplasmic Sperm Injection (ICSI) is a modification of the in vitro fertilisation (“Test-tube baby”) technique where the egg is fertilised outside the body by injecting the sperm directly into it. The embryo(s) are then introduced back into the woman’s uterus in the same way as for IVF.
ICSI is typically suitable for several groups of patients, such as:
- Men with abnormal semen parameters.
- Couples with previous poor fertilisation using IVF.
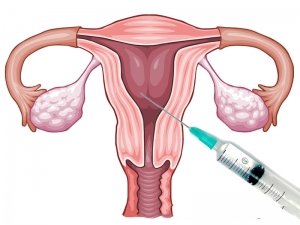
The woman is given fertility drugs to stimulate her ovaries and the development of several mature follicles. Each of these follicles may contain an egg. The recruitment and development of the follicles is regularly monitored by ultrasound scans and blood tests. This is to ensure that the ovarian stimulation is both safe and effective.
Once suitable mature follicles have been produced the eggs are collected using an ultrasound-guided needle. This procedure is performed under sedation/local anaesthetic. The collected eggs are then placed into culture in the embryology laboratory.
The male partner then produces a semen sample, which is then prepared in the laboratory. The aim of the preparation process is to obtain motile, normal looking sperm to be injected into the mature eggs. The Embryologist, using microscope-guided micromanipulators, injects the sperm into suitable eggs.
.
Next morning the eggs are inspected for signs of fertilisation. Those eggs that have normal fertilisation are cultured in the laboratory for another 24 – 48 hours. The development of the embryos is monitored and the patients kept advised of their progress.
The selected embryos are then transfered inside the uterus, through a fine catheter. Typically 2-3 embryos are transferred depending on the patient’s circumstances. The embryo transfer procedure takes no more than half an hour and few women experience any discomfort.
The spare embryos are then assessed in terms of their suitability to be cryopreserved for use, by the couple, in future cycles.
The first ICSI baby was born in 1992 and subsequently many babies have been born through this technique.
Intrauterine insemination
Intra-Uterine Insemination is the introduction of prepared sperm into the uterus in order to improve the chances on pregnancy. IUI-H is applied to couples in which the woman has open fallopian tubes and the male partner has normal semen parameters.
IUI-H involves tracking the progress of the menstrual cycle using ultrasound scans and blood tests. For those women that do not have regular menstrual cycles OI may be employed to help stimulate the production of a mature follicle. The IUI procedure is performed on the day of ovulation.
Once ovulation has been confirmed the male partner produces a semen sample. This semen sample is then prepared in the laboratory by the Embryologist. The aim of the preparation process is to harvest a good number of motile, normal looking sperm. The prepared sperm are then introduced, through a fine catheter, into the woman’s uterus.
The success rate for IUI-H is typically between 10 – 15 % per cycle.
Ovulation Induction (OI)
Ovulation Induction (OI) is the controlled administration of drugs to stimulate the ovaries to produce mature eggs. OI is typically applied to couples in which the woman has irregular menstrual cycles and in which investigations indicate that they may not be ovulating on a regular basis.
Two types of fertility drugs are commonly used in OI:
Clomiphene Citrate: often called Clomid. This drug is an anti-oestrogen and works by stimulating your body into releasing more Follicle Stimulating Hormone (FSH). These higher levels of FSH stimulate the ovaries to produce eggs. Clomid is usually given at the beginning of the menstrual cycle for 5 days. The mature egg is normally released (ovulation) naturally. However, ovulation can also be triggered using by a single injection of human Chorionic Gonadotrophin (hCG).
Gonadotrophin: such as Menopur, Gonal F & Puregon. These drugs are usually given daily to stimulate the development of a single mature follicle. Again, the mature egg is ovulated or the ovulation can be stimulated by hCG.
Ultrasound scans and blood tests are performed to ensure that OI is both safe and effective. Once a mature follicle has been produced you will be advised when the best time for intercourse is in order to help achieve a pregnancy. Typically, couples need only a few months of OI in order to conceive.
Birth Defects
It is important to remember that any medical or surgical treatment has risks, adverse effects and side effects. Couples should be aware that one baby in 20 born worldwide will have a birth defect. There is no clear evidence that infertility medicines, if properly used, increase this risk.
Cancer
Breast Cancer
Of the cancers of the sex organs, breast cancer is most common, occurring in one in eight Barbadian women.
The cause, or aetiology of breast cancer is unknown. Various factors or diseases make breast cancer more likely. Breast cancer in a mother or sister increases the risk, as does some types of non-cancerous (benign) breast lumps.
Breast cancer is more common in infertile women. Some medical research suggests that cigarette smokers are at increased risk.
Monthly breast self-examination is recommended for all women. All lumps should be investigated immediately. Most breast lumps are benign.
There is no screening method for breast cancer, although mammography detects small cancers.
Ovarian Cancer
Occurs in about one in 90 women. Its aetiology is also unknown.
May also occur in families in 10-15% of cases.
It is more common in infertile women.
The oral contraceptive pill decreases the risk of cancer of the ovaries. This remarkable advantage of "The Pill" occurs, not only while the woman takes The Pill, but also for at least five years after stopping The Pill. There is no effective screening method for ovarian cancer. Every woman is advised to have a gynaecological examination and "Pap Smear" every two years to minimise the risk of ovarian, uterine and cervical cancers going undetected.
Cervical Cancer
The lifetime risk of cervical cancer is one in 95 women.
Cervical cancer can be screened by cervical cytology, by the Pap smear.
The aetiology of cervical cancer is related to sexual activity. For this reason, every woman having sexual intercourse should have a Pap smear every two years. An increased number of sexual partners and a papilloma or wart virus infection can also increase the general risk of development of cervical cancer.
A Pap smear every two years, as well as regular gynaecological and breast examinations, are currently the best methods to prevent or detect women's cancers.
Cancer After Infertility and IVF
Doctors have used fertility drugs worldwide since the 1960s, triggering women's ovaries to produce eggs. This approach proved successful in assisting many women with fertility problems to become pregnant and have children.
In the past 20 years the use of fertility drugs has increased markedly following the development of IVF.
Findings of an Australian Study relating to incidence of Cancer and IVF.
Background
The growth in the number of women seeking help to become pregnant provided an opportunity for a study, the largest of its type anywhere in the world.
The study followed up 29,700 women referred to any of ten participating Australian IVF clinics between 1978 and 1993. Of this total:
20,656 women received fertility drugs during IVF (the "treated group")
9,044 women referred for IVF did not end up having fertility drug treatment (the "untreated group").
Depending on the year that women joined an IVF program, the duration of follow-up ranged from one to 22 years, with the majority followed up for five to ten years.
This study was conducted to address concerns that the use of fertility drugs might be associated with an increased risk of cancer.
Study's Aims
To see whether there was any increase in the number of cancers of the breast, ovary and uterus in women on IVF, compared with the number expected among women of the same age, followed up over the same period in the general population.
In the event that there were more of these cancers than predicted, how this might be explained.
Findings
The major finding was that cancers of the breast and ovary were no more common in IVF patients overall than in the general population. Of 29,700 who joined IVF programs, researchers predicted 155 breast cancers and found 143. For ovarian cancer, 13 cases were predicted and 13 were found.
Cancers of the uterus were more common than predicted in untreated IVF patients but were no more common than predicted in the treated group. (Among the 9,044 women in the untreated group, three cases were predicted and seven found).
More women than predicted in the treated group had breast cancer diagnosed in the first year after treatment with fertility drugs. This finding disappeared with time and is discussed below. (Among the 20,656 women, 9 breast cancer cases were predicted in the first year after treatment and 17 were found.)
Women with unexplained infertility had a significantly higher incidence of ovarian and uterine cancer, whether or not they were exposed to fertility drugs, than the general population. (Out of 3,800 women with unexplained infertility, two cases of ovarian cancer were predicted and five were found. With regard to uterine cancer, one case was predicted and five were found).
There was no evidence of any link between the number of treatment cycles or type of fertility drug used and increased cancer incidence. Changes in the types of drugs used in IVF and the amount of ovarian stimulation do not appear to have had an overall impact on cancer in participating women.
Conclusions
The findings provide reassurance that the incidence of breast and ovarian cancers in IVF patients are the same as that for women of the same age in the general population when considered over a five to ten-year period.
The evidence of increased numbers of cancers above, predicted in small numbers of women, in particular subgroups, needs further study. While there was no overall increase, the occurrence of above expected numbers of breast cancer in the first year after treatment is consistent with other research showing a small increase in diagnoses of breast cancer shortly after women give birth. The same effect has been seen in recent users of The Pill and hormone replacement therapy. Possible explanations for this finding after IVF treatment include earlier detection of abnormal breast changes due to close medical supervision, the biological effects of fertility drugs, or both.
Explanations for the relationship between unexplained infertility and the increase in ovarian and uterine cancers are harder to come by but research is continuing. In a bid to explore such issues, the research team has started a more detailed investigation of about 700 IVF patients, some of whom have cancer and others who do not.
Summary
The risk of cancers in women is best reduced through regular women's health check-ups, breast examination, Pap smears and gynaecological examinations - and by the use, at other times, of The Pill. The most effective way of minimising the risk of breast and ovarian cancers in infertile women is to help them to have a baby. However, the study does reinforce the importance of women having medical check-ups at regular intervals after fertility treatment.